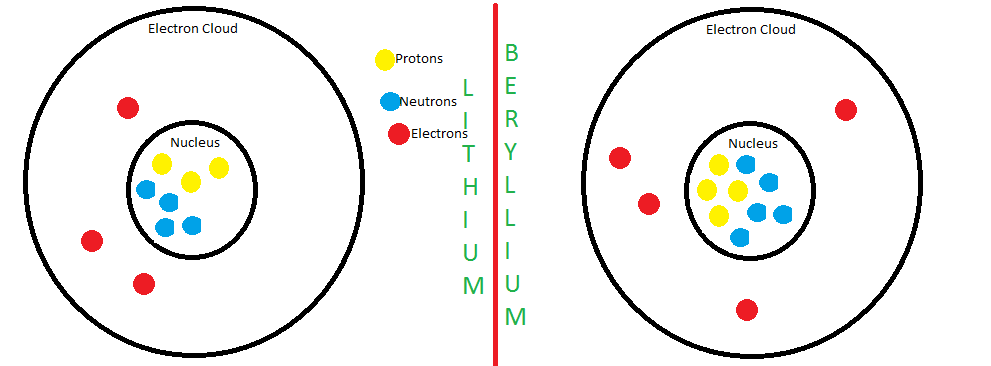
Atom Patterns
***Part 2
***FOR NEUTRAL (A=P=E) Atomic number=protons=electrons
(M-A=N) Mass-Atomic number=neutrons
-Protons and neutrons make up the mass of an atom and are found in the nucleus.
-Electrons do not contribute significant mass to an atom but do make up the volume of an atom since they are found in the electron cloud.
-The overall charge of the nucleus is positive because of the positive protons and neutral neutrons.
-The charge of the electron cloud is negative because of the negative electrons.
-The attraction between the positive charge of the nucleus and the negative charge of the electron cloud holds the atom together.
-Protons, neutrons, and electrons are subatomic particles. Atoms are different from each other because the numbers of the subatomic particles are different.
-To measure the overall charge, count up the positive atoms and the negative atoms and add those together.
-As long as the number of protons and electrons are equal, the atom is without charge, or neutral.
-Atomic number is the same thing as a proton.
***FOR NEUTRAL (A=P=E) Atomic number=protons=electrons
(M-A=N) Mass-Atomic number=neutrons
-Protons and neutrons make up the mass of an atom and are found in the nucleus.
-Electrons do not contribute significant mass to an atom but do make up the volume of an atom since they are found in the electron cloud.
-The overall charge of the nucleus is positive because of the positive protons and neutral neutrons.
-The charge of the electron cloud is negative because of the negative electrons.
-The attraction between the positive charge of the nucleus and the negative charge of the electron cloud holds the atom together.
-Protons, neutrons, and electrons are subatomic particles. Atoms are different from each other because the numbers of the subatomic particles are different.
-To measure the overall charge, count up the positive atoms and the negative atoms and add those together.
-As long as the number of protons and electrons are equal, the atom is without charge, or neutral.
-Atomic number is the same thing as a proton.
1) Explain why the charge is 0 (zero) for each of the four atoms that you built on the diagrams.
Because since there is an equal amount of protons and electrons on each element, they cancel eachother out, so therefore, they have no charge.
2) Rewrite the following statement so that it is TRUE: The number of protons and neutrons for these atoms increase sequentially by one.
The number of protons and electrons for these atoms increase sequentially by one.
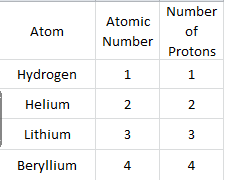
3) Use a periodic table to look up the atomic numbers for hydrogen, helium, lithium, and beryllium. Complete the table below and answer the follow-up questions below the table.
Because since there is an equal amount of protons and electrons on each element, they cancel eachother out, so therefore, they have no charge.
2) Rewrite the following statement so that it is TRUE: The number of protons and neutrons for these atoms increase sequentially by one.
The number of protons and electrons for these atoms increase sequentially by one.
3) Use a periodic table to look up the atomic numbers for hydrogen, helium, lithium, and beryllium. Complete the table below and answer the follow-up questions below the table.
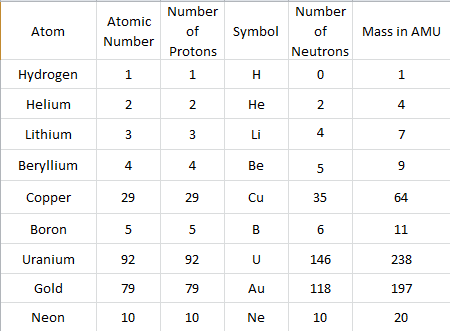
Use the trend that you see in the table above to predict how many protons will be found in an atom of:
Copper (Cu) 29
Boron (B) 5
Uranium (U) 92
Gold (Au) 79
Neon (Ne) 10
What statement can be made about an atom's atomic number and the atom's number of protons?
The atomic number and the atom's number of protons are equal.
4) Use the same group of elements (copper, boron, uranium, gold, and neon) to predict the number of electrons found in a single NEUTRAL atom (an atom without charge).
Copper (Cu) 29
Boron (B) 5
Uranium (U) 92
Gold (Au) 79
Neon (Ne) 10
What statement can be made about a neutral atom's number of electrons compared to the atoms number of protons?
If the atom is neutral in charge, then the number of electrons and protons has to be the same.
5) If we know an atom's mass in amu and we know the number of protons of the atom, can we calculate the remaining number of neutrons that contribute mass using a simple subtraction? yes
Look at the example, using the atoms that you built on the diagrams, then complete the rest of the chart.
Copper (Cu) 29
Boron (B) 5
Uranium (U) 92
Gold (Au) 79
Neon (Ne) 10
What statement can be made about an atom's atomic number and the atom's number of protons?
The atomic number and the atom's number of protons are equal.
4) Use the same group of elements (copper, boron, uranium, gold, and neon) to predict the number of electrons found in a single NEUTRAL atom (an atom without charge).
Copper (Cu) 29
Boron (B) 5
Uranium (U) 92
Gold (Au) 79
Neon (Ne) 10
What statement can be made about a neutral atom's number of electrons compared to the atoms number of protons?
If the atom is neutral in charge, then the number of electrons and protons has to be the same.
5) If we know an atom's mass in amu and we know the number of protons of the atom, can we calculate the remaining number of neutrons that contribute mass using a simple subtraction? yes
Look at the example, using the atoms that you built on the diagrams, then complete the rest of the chart.