Bag Lab
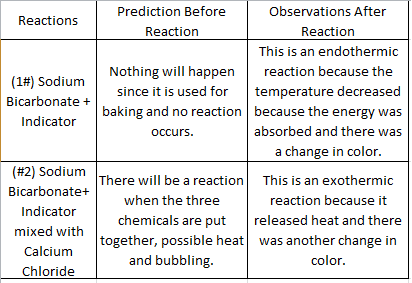
1) How do the results of each reaction compare with your predictions for that reaction?
2) What was/were the observable chemical indications that lead you to believe a chemical reaction occurred when you mixed sodium bicarbonate and indicator solution #1?
3) What was/were the observable chemical indications that lead you to believe a chemical reaction occurred when you mixed sodium bicarbonate and indicator solution #2?
4) Gas formation is one possible indicator of a chemical change. Is there ever an instance when gas formation could indicate a physical change in a substance rather than a chemical change?
5) The temperature of two separate solutions in found to be 20 C. When the solutions are mixed, a precipitate forms and the temperature changes to 18 C. Classify the type of change in energy and explain your answer.
2) What was/were the observable chemical indications that lead you to believe a chemical reaction occurred when you mixed sodium bicarbonate and indicator solution #1?
3) What was/were the observable chemical indications that lead you to believe a chemical reaction occurred when you mixed sodium bicarbonate and indicator solution #2?
4) Gas formation is one possible indicator of a chemical change. Is there ever an instance when gas formation could indicate a physical change in a substance rather than a chemical change?
5) The temperature of two separate solutions in found to be 20 C. When the solutions are mixed, a precipitate forms and the temperature changes to 18 C. Classify the type of change in energy and explain your answer.