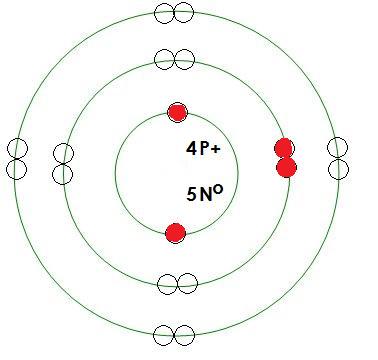
Modeling Electron Clouds
-The layer that fills with electrons first is that layer CLOSEST to the nucleus, then the second layer and so on
-There can be up to 7 layers
-Each layer is an energy layer.
-Up to 2 in the 1st energy level.
-Up to 8 in the 2, 3, 4th energy levels.
-Levels in the periodic table determine how many energy levels are in the atom.
**ELEMENTS
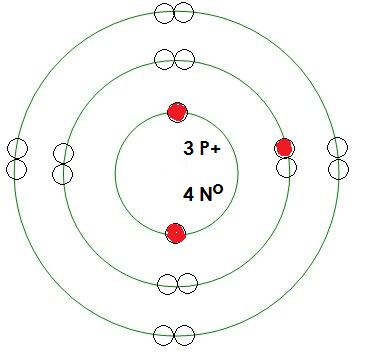
-Lithium
Mass= 7 amu
Protons= 3
Neutrons= 4
Electrons= 3
How many positive charges in this atom? 3
How many negative charges in this atom? 3
What energy level is being filled in this atom? 2
Is this energy level full? no
-Beryllium
Mass= 9 amu
Protons= 4
Neutrons= 5
Electrons= 4
How many positive charges in this atom? 4
How many negative charges in this atom? 4
What energy level is being filled in this atom? 2
Is this energy level full? no
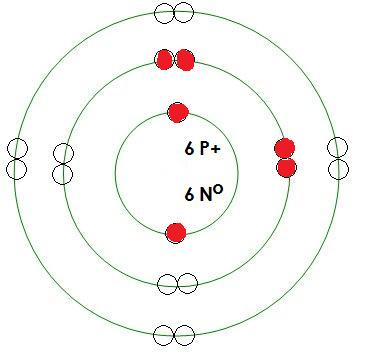
-Carbon
Mass= 12 amu
Protons= 6
Neutrons= 6
Electrons= 6
How many positive charges in this atom? 6
How many negative charges in this atom? 6
What energy level is being filled in this atom? 2
Is this energy level full? no
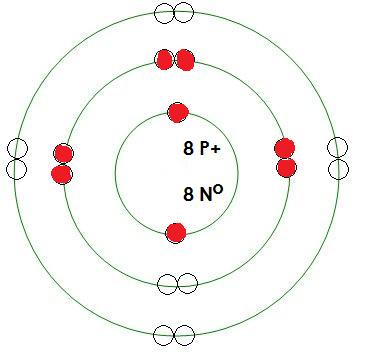
-Oxygen
Mass= 16 amu
Protons= 8
Neutrons= 8
Electrons= 8
How many positive charges in this atom? 8
How many negative charges in this atom? 8
What energy level is being filled in this atom? 2
Is this energy level full? no
-Neon
Mass= 20 amu
Protons= 10
Neutrons= 10
Electrons= 10
How many positive charges in this atom? 10
How many negative charges in this atom? 10
What energy level is being filled in this atom? 2
Is this energy level full? yes
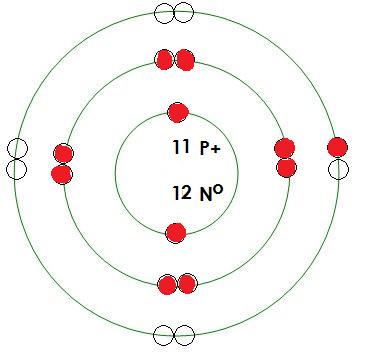
-Sodium
Mass= 23 amu
Protons= 11
Neutrons= 12
Electrons= 11
How many positive charges in this atom? 11
How many negative charges in this atom? 11
What energy level is being filled in this atom? 2
Is this energy level full? no
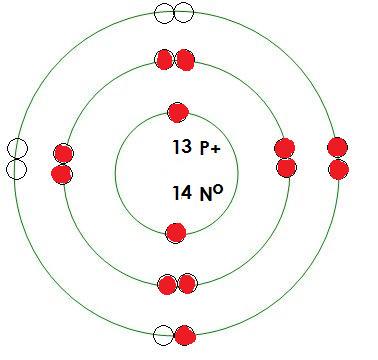
-Aluminum
Mass= 27 amu
Protons= 13
Neutrons= 14
Electrons= 13
How many positive charges in this atom? 13
How many negative charges in this atom? 13
What energy level is being filled in this atom? 3
Is this energy level full? no
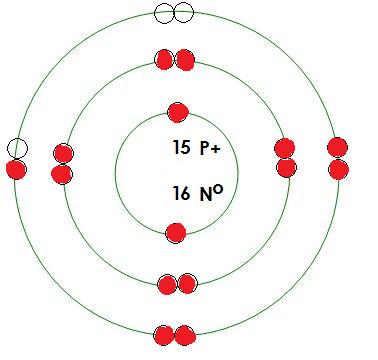
-Phosphorus
Mass= 31 amu
Protons= 15
Neutrons= 16
Electrons= 15
How many positive charges in this atom? 15
How many negative charges in this atom? 15
What energy level is being filled in this atom? 3
Is this energy level full? no
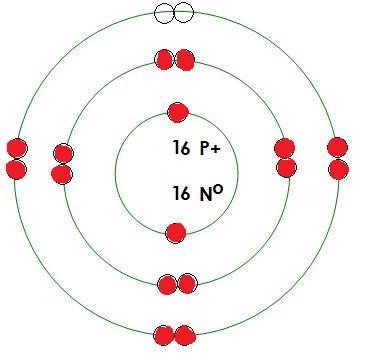
-Sulfur
Mass= 32 amu
Protons= 16
Neutrons= 16
Electrons= 16
How many positive charges in this atom? 16
How many negative charges in this atom? 16
What energy level is being filled in this atom? 3
Is this energy level full? no
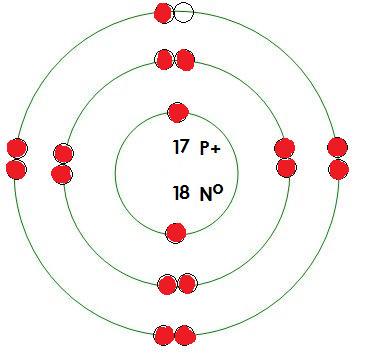
-Chlorine
Mass= 35 amu
Protons= 17
Neutrons= 18
Electrons= 17
How many positive charges in this atom? 17
How many negative charges in this atom? 17
What energy level is being filled in this atom? 3
Is this energy level full? no
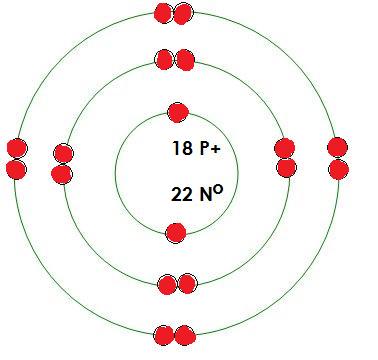
-Argon
Mass= 40 amu
Protons= 18
Neutrons= 22
Electrons= 18
How many positive charges in this atom? 18
How many negative charges in this atom? 18
What energy level is being filled in this atom? 3
Is this energy level full? yes
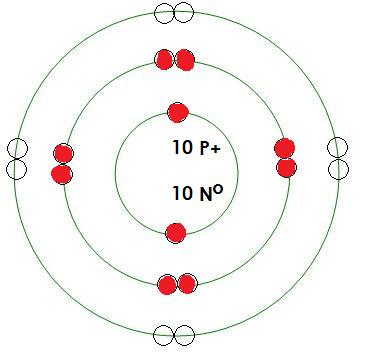
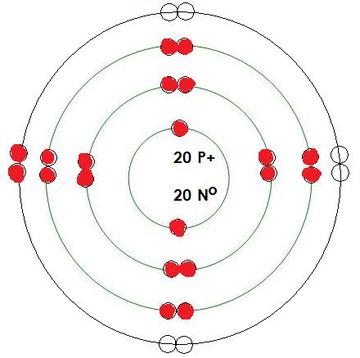
-Calcium
Mass= 40 amu
Protons= 20
Neutrons= 20
Electrons= 20
How many positive charges in this atom? 20
How many negative charges in this atom? 20
What energy level is being filled in this atom? 4
Is this energy level full? no
-There can be up to 7 layers
-Each layer is an energy layer.
-Up to 2 in the 1st energy level.
-Up to 8 in the 2, 3, 4th energy levels.
-Levels in the periodic table determine how many energy levels are in the atom.
**ELEMENTS
-Lithium
Mass= 7 amu
Protons= 3
Neutrons= 4
Electrons= 3
How many positive charges in this atom? 3
How many negative charges in this atom? 3
What energy level is being filled in this atom? 2
Is this energy level full? no
-Beryllium
Mass= 9 amu
Protons= 4
Neutrons= 5
Electrons= 4
How many positive charges in this atom? 4
How many negative charges in this atom? 4
What energy level is being filled in this atom? 2
Is this energy level full? no
-Carbon
Mass= 12 amu
Protons= 6
Neutrons= 6
Electrons= 6
How many positive charges in this atom? 6
How many negative charges in this atom? 6
What energy level is being filled in this atom? 2
Is this energy level full? no
-Oxygen
Mass= 16 amu
Protons= 8
Neutrons= 8
Electrons= 8
How many positive charges in this atom? 8
How many negative charges in this atom? 8
What energy level is being filled in this atom? 2
Is this energy level full? no
-Neon
Mass= 20 amu
Protons= 10
Neutrons= 10
Electrons= 10
How many positive charges in this atom? 10
How many negative charges in this atom? 10
What energy level is being filled in this atom? 2
Is this energy level full? yes
-Sodium
Mass= 23 amu
Protons= 11
Neutrons= 12
Electrons= 11
How many positive charges in this atom? 11
How many negative charges in this atom? 11
What energy level is being filled in this atom? 2
Is this energy level full? no
-Aluminum
Mass= 27 amu
Protons= 13
Neutrons= 14
Electrons= 13
How many positive charges in this atom? 13
How many negative charges in this atom? 13
What energy level is being filled in this atom? 3
Is this energy level full? no
-Phosphorus
Mass= 31 amu
Protons= 15
Neutrons= 16
Electrons= 15
How many positive charges in this atom? 15
How many negative charges in this atom? 15
What energy level is being filled in this atom? 3
Is this energy level full? no
-Sulfur
Mass= 32 amu
Protons= 16
Neutrons= 16
Electrons= 16
How many positive charges in this atom? 16
How many negative charges in this atom? 16
What energy level is being filled in this atom? 3
Is this energy level full? no
-Chlorine
Mass= 35 amu
Protons= 17
Neutrons= 18
Electrons= 17
How many positive charges in this atom? 17
How many negative charges in this atom? 17
What energy level is being filled in this atom? 3
Is this energy level full? no
-Argon
Mass= 40 amu
Protons= 18
Neutrons= 22
Electrons= 18
How many positive charges in this atom? 18
How many negative charges in this atom? 18
What energy level is being filled in this atom? 3
Is this energy level full? yes
-Calcium
Mass= 40 amu
Protons= 20
Neutrons= 20
Electrons= 20
How many positive charges in this atom? 20
How many negative charges in this atom? 20
What energy level is being filled in this atom? 4
Is this energy level full? no
PART II: Valence Shell and Valence Electrons
Valence Shell: outer energy level
Valence Electrons: how many electrons there's in the outer layer
1) How many valence electrons are found in a neutral atom of beryllium? two
2) How many valence electrons are found in a neutral atom of nitrogen? five
3) How many valence electrons are found in a neutral atom of oxygen? 6
4) How many valence electrons are found in a neutral atom of neon? 8
5) Is the valence shell of neon full? yes
6) How many valence electrons are found in a neutral atom of sodium? 1
7) How many valence electrons are found in a neutral atom of magnesium? 2
8) How many valence electrons are found in a neutral atom of chlorine? 7
9) How many valence electrons are found in a neutral atom of argon? 8
10) Is the valence shell of argon full? yes
Valence Shell: outer energy level
Valence Electrons: how many electrons there's in the outer layer
1) How many valence electrons are found in a neutral atom of beryllium? two
2) How many valence electrons are found in a neutral atom of nitrogen? five
3) How many valence electrons are found in a neutral atom of oxygen? 6
4) How many valence electrons are found in a neutral atom of neon? 8
5) Is the valence shell of neon full? yes
6) How many valence electrons are found in a neutral atom of sodium? 1
7) How many valence electrons are found in a neutral atom of magnesium? 2
8) How many valence electrons are found in a neutral atom of chlorine? 7
9) How many valence electrons are found in a neutral atom of argon? 8
10) Is the valence shell of argon full? yes