Warm-Ups
4/12/12
What is matter? Physical substance in general, as distinct from mind and spirit; (in physics) that which occupies space and possesses rest mass, esp. as distinct from energy.
4/13/12
What is an atom? The smallest particle of an element
4/16/12
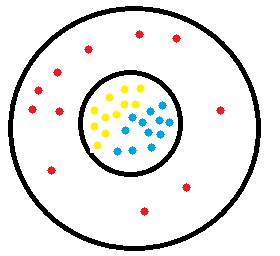
Where is the mass of an atom found? In the nucleus of the atom
What is the mass of this atom? 6 amu
4/17/12
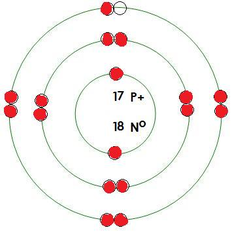
1) Charge? neutral
2) Mass? 7 amu
3) Atomic Number? 6
4) Element? Carbon
4/18/12
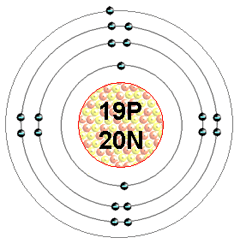
1)Protons? 11
2) Electrons? 11
3) Neutrons? 12
4) Draw atom
What is matter? Physical substance in general, as distinct from mind and spirit; (in physics) that which occupies space and possesses rest mass, esp. as distinct from energy.
4/13/12
What is an atom? The smallest particle of an element
4/16/12
Where is the mass of an atom found? In the nucleus of the atom
What is the mass of this atom? 6 amu
4/17/12
1) Charge? neutral
2) Mass? 7 amu
3) Atomic Number? 6
4) Element? Carbon
4/18/12
1)Protons? 11
2) Electrons? 11
3) Neutrons? 12
4) Draw atom
4/19/12
-Chlorine
mass: 35
electrons: 17
protons: 17
neutrons: 18
energy level: 3, not full
-Chlorine
mass: 35
electrons: 17
protons: 17
neutrons: 18
energy level: 3, not full
4/20/12
-Potassium has 1 valance electrons
-Potassium has 1 valance electrons
4/23/12
Magnesium:
# of valance electrons: 2
Stable? No, it's not stable, it will lose electrons (1, 2, or 3 loses, 4 shares, 5, 6, or 7 will gain) therefore, the charge will be positive
2/25/12
1) how many elements are in the reaction above?
three
2) how many atoms are in the products of the reaction?
36
3) does this equation violate the law of conservation of matter?
no
5/1/12
Groups or families on the periodic table have what in common? Valance electrons
Periods on the periodic table have what in common? Energy levels
5/2/12
-What is the atomic number of the element above? 3
-What is the amu of the element? 7 amu
-Is the element an ion? no because the number of protons and neutrons are equal.
5/3/12
-Bromnie
-Zircronium
5/7/12
-How many elements and atoms are in each of the molecules?
-three elements, 13 total atoms
-two elements, 15 total atoms
-three elements, 7 total atoms
5/8/12
-5HIO3 + 4FeI2 +25HCl --> 4FeCl3 + 13ICl +15H20
5/10/12
- 2H2SO4 + Pb(OH)4 --> Pb(SO4)2 + 4H2O
-C12H22O11 + 8KClO3 --> 12CO2 + 11H2O + 8KCl
5/11/12
-Ca3(PO4)2 + 2H2SP4 --> 2CaSO4 + Ca(H2PO4)2
5/15/12
-2 Fe + 3 H2SO4 --> 1 Fe2(SO4)3 + 3 H2
Magnesium:
# of valance electrons: 2
Stable? No, it's not stable, it will lose electrons (1, 2, or 3 loses, 4 shares, 5, 6, or 7 will gain) therefore, the charge will be positive
2/25/12
1) how many elements are in the reaction above?
three
2) how many atoms are in the products of the reaction?
36
3) does this equation violate the law of conservation of matter?
no
5/1/12
Groups or families on the periodic table have what in common? Valance electrons
Periods on the periodic table have what in common? Energy levels
5/2/12
-What is the atomic number of the element above? 3
-What is the amu of the element? 7 amu
-Is the element an ion? no because the number of protons and neutrons are equal.
5/3/12
-Bromnie
-Zircronium
5/7/12
-How many elements and atoms are in each of the molecules?
-three elements, 13 total atoms
-two elements, 15 total atoms
-three elements, 7 total atoms
5/8/12
-5HIO3 + 4FeI2 +25HCl --> 4FeCl3 + 13ICl +15H20
5/10/12
- 2H2SO4 + Pb(OH)4 --> Pb(SO4)2 + 4H2O
-C12H22O11 + 8KClO3 --> 12CO2 + 11H2O + 8KCl
5/11/12
-Ca3(PO4)2 + 2H2SP4 --> 2CaSO4 + Ca(H2PO4)2
5/15/12
-2 Fe + 3 H2SO4 --> 1 Fe2(SO4)3 + 3 H2