Reactivity
Part III: Reactivity
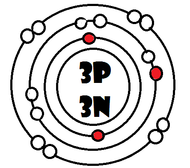
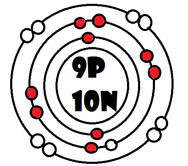
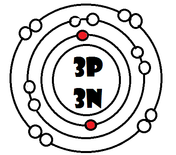
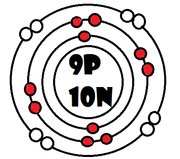
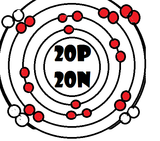
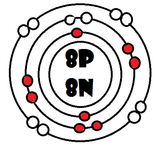
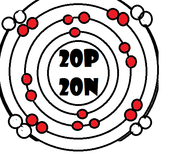
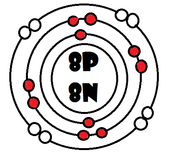
As it turns our, the key number for the valence shell is the number *. The number 2 is also a key number, but only for the atoms Hydrogen, Helium, Lithium, Beryllium, and Boron. So what makes 8, and sometimes 2, key numbers?
Scientists learned a simple rule of thumb about atoms. they tend to combine so that their valence shells are full of electrons. This is called the octet rule. (note: the root oct=8 in the word octet).
This means two things:
1) If a neutral atom's valence shell is full, then the atom will not react with other atoms.
2) If a neutral atom's valence shell has space available, then the atom will react with other atoms based on the number of electrons found in the valence shell.
-Some atoms only need 1, 2, or 3 more electrons to fill the valence shell. These electrons come from other atoms when they react.
-Some atoms need to remove 1, 2, or 3 electrons to have the energy layer below become the valence shell that is already full. The removed electrons are accepted by other atoms.
-Some atoms tend to react with other atoms in a way electrons are shared rather than giving up or receiving electrons.
As it turns our, the key number for the valence shell is the number *. The number 2 is also a key number, but only for the atoms Hydrogen, Helium, Lithium, Beryllium, and Boron. So what makes 8, and sometimes 2, key numbers?
Scientists learned a simple rule of thumb about atoms. they tend to combine so that their valence shells are full of electrons. This is called the octet rule. (note: the root oct=8 in the word octet).
This means two things:
1) If a neutral atom's valence shell is full, then the atom will not react with other atoms.
2) If a neutral atom's valence shell has space available, then the atom will react with other atoms based on the number of electrons found in the valence shell.
-Some atoms only need 1, 2, or 3 more electrons to fill the valence shell. These electrons come from other atoms when they react.
-Some atoms need to remove 1, 2, or 3 electrons to have the energy layer below become the valence shell that is already full. The removed electrons are accepted by other atoms.
-Some atoms tend to react with other atoms in a way electrons are shared rather than giving up or receiving electrons.
REACTION 2
Lithium atom
|
Fluorine atom
|
Lithium ion
|
Fluorine ion
|
REACTION 3
Calcium atom
|
Oxygen atom
|
Calcium ion
|
Oxygen ion
|